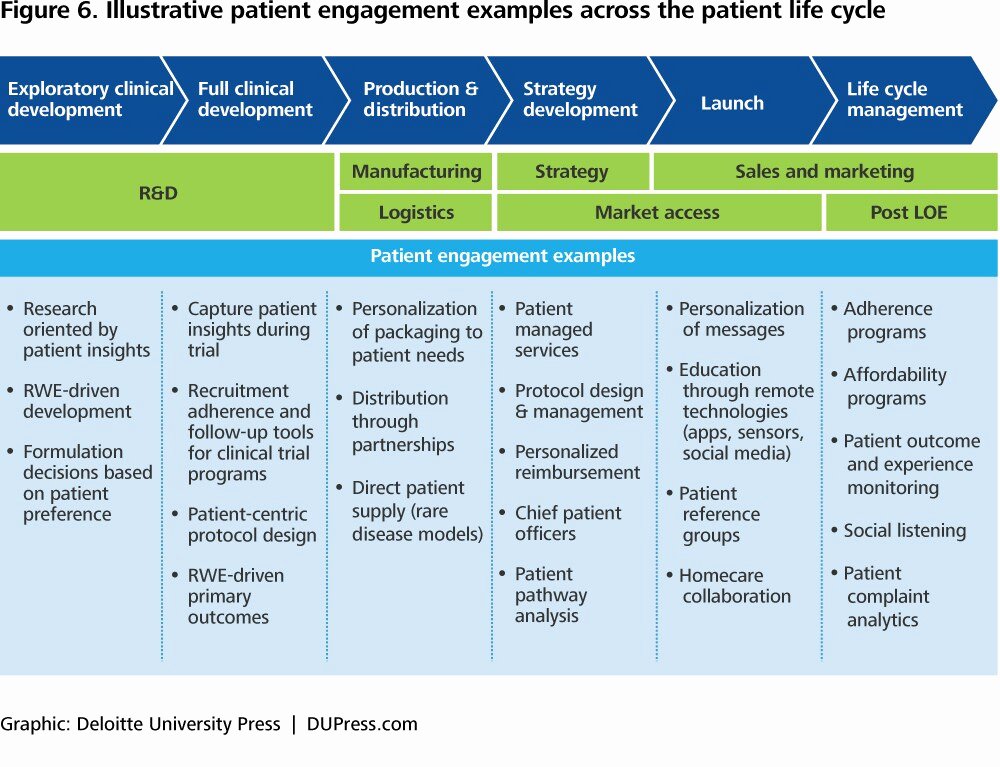
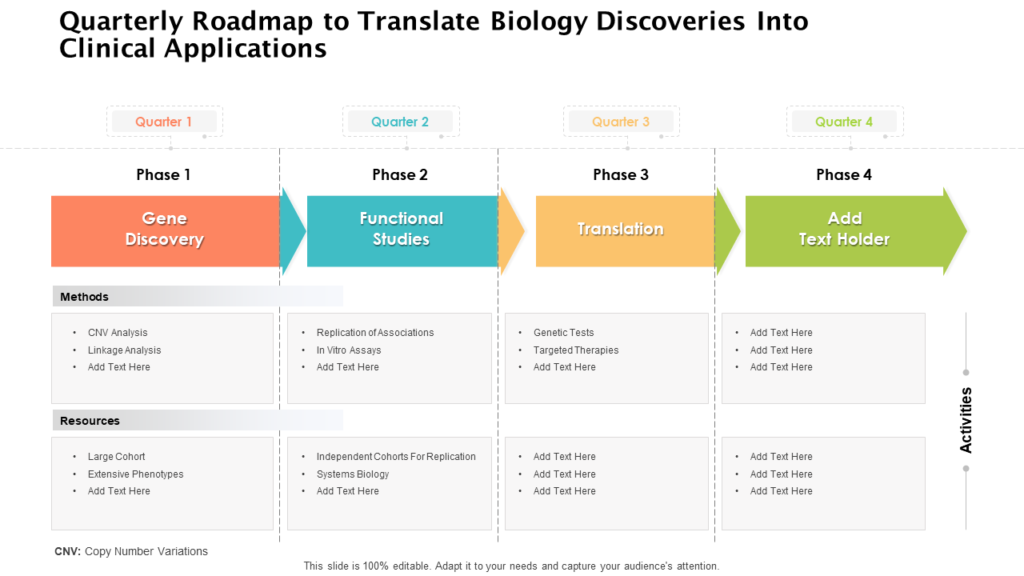
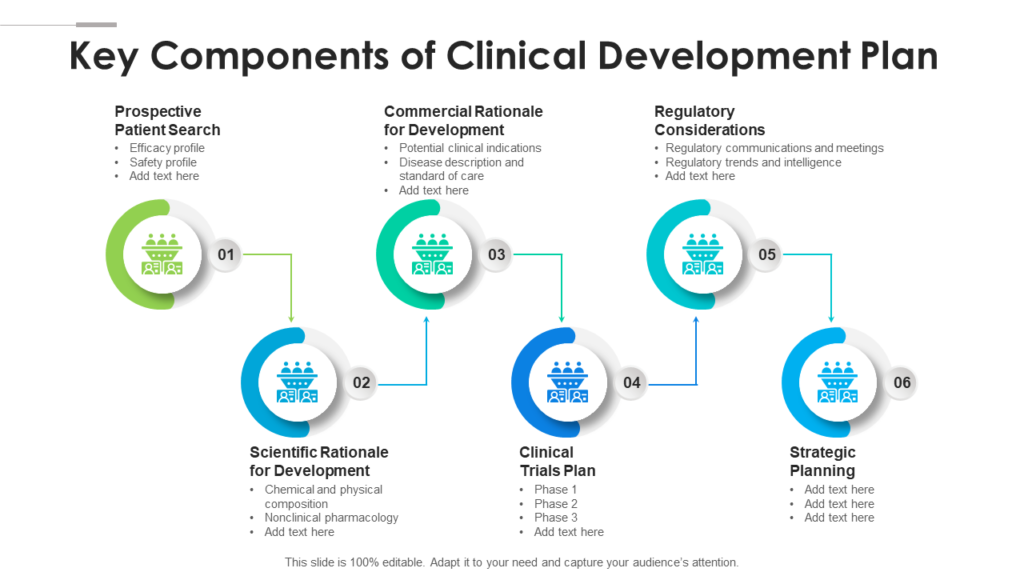
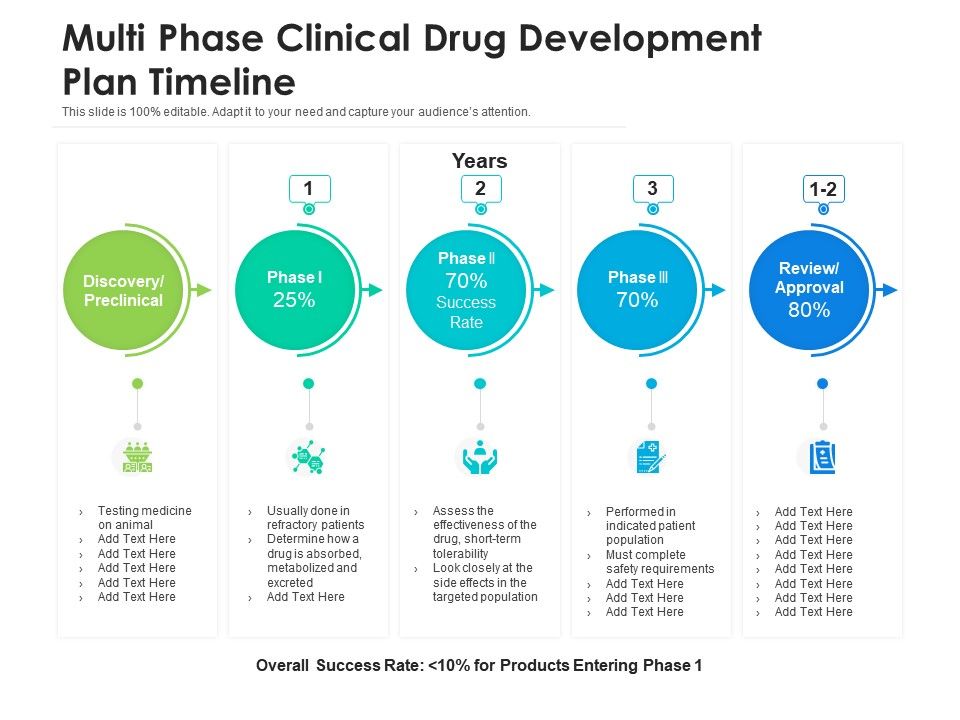
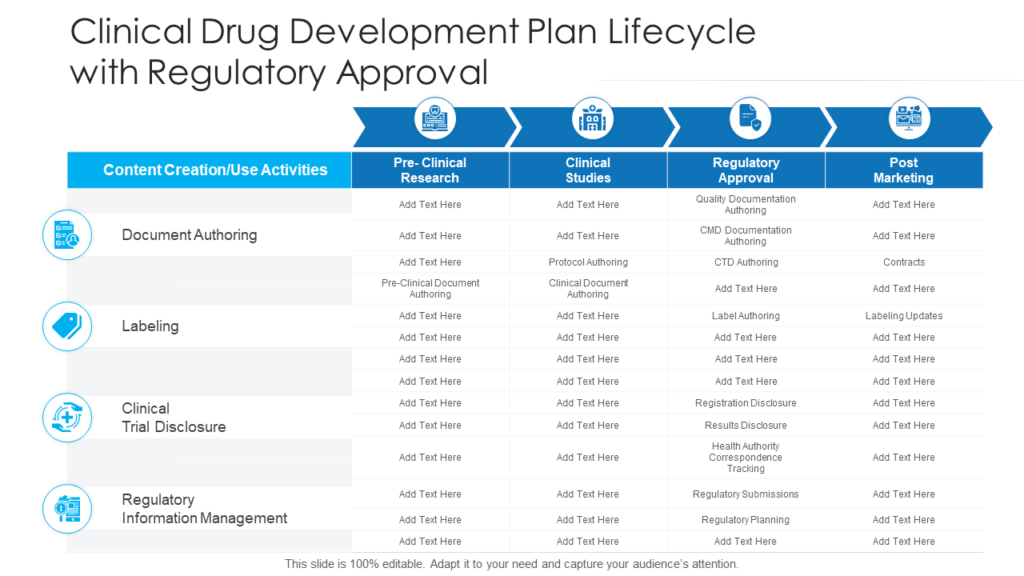
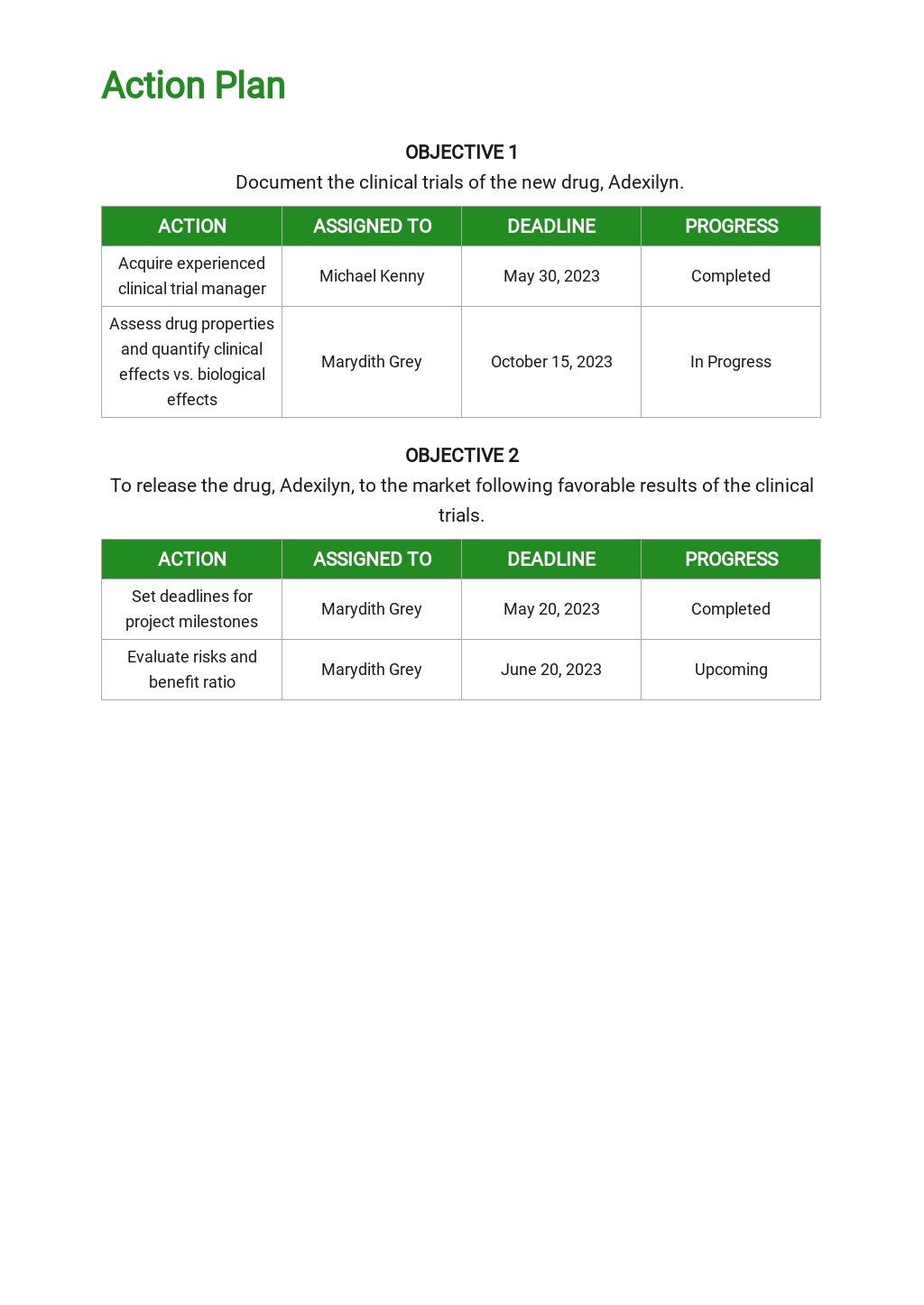
Clinical Development Plan Template
Clinical Development Plan Template - Web learn how to create a cdp for eu mdr compliance and strategic planning of medical devices. Find out what the cdp should cover, how it differs from the cer, and what guidance is available. Web learn how to create a comprehensive and strategic clinical development plan for your new pharmaceutical or biotechnology product. Find out the differences between pilot, pivotal, and pmcf studies and how to present them in the cep. Web learn how to create a strategic roadmap for advancing a new compound from the lab to the market, including a target product profile, a regulatory strategy, a clinical development plan and a commercial plan. See a suggested document structure, regulatory considerations, and tips for new and existing devices. A cdp can help optimize efficiency, control costs, plan timelines, and maximize the probability of success for a new drug program. It explains the aims, methods and procedures of pmcf and the relevant sections of the template. The vision is transformed into distinct implementation phases and discrete steps, called clinical studies, each with well defined milestones and deliverables. This white paper provides an overview of the key elements and challenges of drug development, with examples and resources. A cdp can help optimize efficiency, control costs, plan timelines, and maximize the probability of success for a new drug program. This white paper provides an overview of the key elements and challenges of drug development, with examples and resources. Web learn how to create a comprehensive and strategic clinical development plan for your new pharmaceutical or biotechnology product. It explains the aims, methods and procedures of pmcf and the relevant sections of the template. See a suggested document structure, regulatory considerations, and tips for new and existing devices. This guide covers the key components, such as prospective patient package insert, scientific and commercial rationale, clinical trials plan, regulatory considerations, and strategic planning. The vision is transformed into distinct implementation phases and discrete steps, called clinical studies, each with well defined milestones and deliverables. It covers various aspects of the clinical evaluation, such as device description, clinical literature review, clinical investigations,. Web this document is a harmonised template for notified bodies to document their assessment of the clinical evidence and related documents presented by manufacturers in accordance with the medical device regulation (eu) 2017/745. Web learn how to create a cdp for eu mdr compliance and strategic planning of medical devices. This white paper provides an overview of the key elements and challenges of drug development, with examples and resources. Web learn how to plan and document your medical device's clinical evaluation according to the mdr and iso 14155:2020. Web learn how to create a cdp for eu mdr compliance and strategic planning of medical devices. It explains the aims, methods and procedures of pmcf and the relevant sections of the template. The vision is transformed into distinct implementation phases and discrete steps, called clinical studies, each with well defined milestones and deliverables. A cdp can help optimize efficiency, control costs, plan timelines, and maximize the probability of success for a new drug program. It covers various aspects of the clinical evaluation, such as device description, clinical literature review, clinical investigations,. Web learn how to create a comprehensive and strategic clinical development plan for your new pharmaceutical or biotechnology product. See a suggested document structure, regulatory considerations, and tips for new and existing devices. Web learn how to comply with the mdr requirement to establish clinical development plans (cdp) for new and existing medical devices. Web learn how to comply with the mdr requirement to establish clinical development plans (cdp) for new and existing medical devices. This guide covers the key components, such as prospective patient package insert, scientific and commercial rationale, clinical trials plan, regulatory considerations, and strategic planning. Find out what the cdp should cover, how it differs from the cer, and what. Web learn how to plan and document your medical device's clinical evaluation according to the mdr and iso 14155:2020. Web learn how to create a comprehensive and strategic clinical development plan for your new pharmaceutical or biotechnology product. The vision is transformed into distinct implementation phases and discrete steps, called clinical studies, each with well defined milestones and deliverables. It. It covers various aspects of the clinical evaluation, such as device description, clinical literature review, clinical investigations,. The vision is transformed into distinct implementation phases and discrete steps, called clinical studies, each with well defined milestones and deliverables. This guide covers the key components, such as prospective patient package insert, scientific and commercial rationale, clinical trials plan, regulatory considerations, and. Web learn how to create a strategic roadmap for advancing a new compound from the lab to the market, including a target product profile, a regulatory strategy, a clinical development plan and a commercial plan. Web learn how to plan and document your medical device's clinical evaluation according to the mdr and iso 14155:2020. Web this document is a harmonised. Web learn how to create a strategic roadmap for advancing a new compound from the lab to the market, including a target product profile, a regulatory strategy, a clinical development plan and a commercial plan. Web this document is a harmonised template for notified bodies to document their assessment of the clinical evidence and related documents presented by manufacturers in. Web learn how to create a cdp for eu mdr compliance and strategic planning of medical devices. Web learn how to plan and document your medical device's clinical evaluation according to the mdr and iso 14155:2020. It explains the aims, methods and procedures of pmcf and the relevant sections of the template. This white paper provides an overview of the. See a suggested document structure, regulatory considerations, and tips for new and existing devices. A cdp can help optimize efficiency, control costs, plan timelines, and maximize the probability of success for a new drug program. Find out the differences between pilot, pivotal, and pmcf studies and how to present them in the cep. The vision is transformed into distinct implementation. This white paper provides an overview of the key elements and challenges of drug development, with examples and resources. Web this document is a harmonised template for notified bodies to document their assessment of the clinical evidence and related documents presented by manufacturers in accordance with the medical device regulation (eu) 2017/745. See a suggested document structure, regulatory considerations, and. See a suggested document structure, regulatory considerations, and tips for new and existing devices. It covers various aspects of the clinical evaluation, such as device description, clinical literature review, clinical investigations,. Find out the differences between pilot, pivotal, and pmcf studies and how to present them in the cep. This guide covers the key components, such as prospective patient package. Web learn how to comply with the mdr requirement to establish clinical development plans (cdp) for new and existing medical devices. Web learn how to create a cdp for eu mdr compliance and strategic planning of medical devices. It covers various aspects of the clinical evaluation, such as device description, clinical literature review, clinical investigations,. The vision is transformed into. It covers various aspects of the clinical evaluation, such as device description, clinical literature review, clinical investigations,. The vision is transformed into distinct implementation phases and discrete steps, called clinical studies, each with well defined milestones and deliverables. This guide covers the key components, such as prospective patient package insert, scientific and commercial rationale, clinical trials plan, regulatory considerations, and strategic planning. Find out the differences between pilot, pivotal, and pmcf studies and how to present them in the cep. This white paper provides an overview of the key elements and challenges of drug development, with examples and resources. Web learn how to create a comprehensive and strategic clinical development plan for your new pharmaceutical or biotechnology product. Web learn how to create a cdp for eu mdr compliance and strategic planning of medical devices. See a suggested document structure, regulatory considerations, and tips for new and existing devices. Web learn how to plan and document your medical device's clinical evaluation according to the mdr and iso 14155:2020. It explains the aims, methods and procedures of pmcf and the relevant sections of the template. Find out what the cdp should cover, how it differs from the cer, and what guidance is available. Web this document is a harmonised template for notified bodies to document their assessment of the clinical evidence and related documents presented by manufacturers in accordance with the medical device regulation (eu) 2017/745.Clinical Development Plan Template
Roadmap For Clinical Development Plan Implementation Presentation
Clinical Development Plan Template
Clinical Development Plan Template Google Docs, Word, Apple Pages
Top 20 PowerPoint Templates to Create a Clinical Development Plan
Clinical Development Plan Template in Google Docs, Pages, Word
Top 20 PowerPoint Templates to Create a Clinical Development Plan
Clinical Development Plan Template
Top 20 PowerPoint Templates to Create a Clinical Development Plan
Clinical Development Plan Template
Web Learn How To Create A Strategic Roadmap For Advancing A New Compound From The Lab To The Market, Including A Target Product Profile, A Regulatory Strategy, A Clinical Development Plan And A Commercial Plan.
Web Learn How To Comply With The Mdr Requirement To Establish Clinical Development Plans (Cdp) For New And Existing Medical Devices.
A Cdp Can Help Optimize Efficiency, Control Costs, Plan Timelines, And Maximize The Probability Of Success For A New Drug Program.
Related Post: